x
k
l
a
r
c
h
e
m
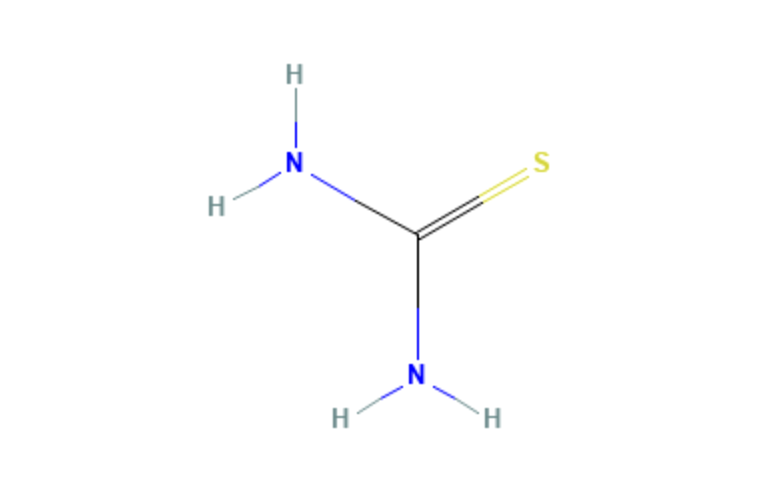
Thio Urea
Chemical Product
Thio Urea
Physical and Chemical Properties:
| Properties | |
|---|---|
| Appearance | White crystalline solid |
| Odor | Slight sulfur odor |
| Molecular Formula | CH4N2S |
| Molecular Weight | 76.12 g/mol |
| Melting Point | 171–173 °C |
| Density | 1.37 g/cm³ |
| Solubility | Soluble in water, ethanol, and acetone |
| Flash Point | Not applicable |
| Chemical Stability | Stable under normal conditions, but decomposes under acidic conditions |
Thio Urea is an organic sulfur compound used in the production of pharmaceuticals, pesticides, and as a stabilizer in photographic chemicals.
- Chemical Manufacturing: Used in the production of pesticides, herbicides, and other agrochemicals.
- Pharmaceuticals: Applied in the synthesis of various drugs and as an intermediate in chemical synthesis.
- Photographic Chemicals: Used in photographic development and as a stabilizer in photographic solutions.
- Laboratory Reagents: Used in chemical analysis and research.
Physical and Chemical Properties:
| Properties | Data |
|---|---|
| Appearance | White crystalline solid |
| Odor | Slight sulfur odor |
| Molecular Formula | CH4N2S |
| Molecular Weight | 76.12 g/mol |
| Melting Point | 171–173 °C |
| Density | 1.37 g/cm³ |
| Solubility | Soluble in water, ethanol, and acetone |
| Flash Point | Not applicable |
| Chemical Stability | Stable under normal conditions, but decomposes under acidic conditions |