x
k
l
a
r
c
h
e
m
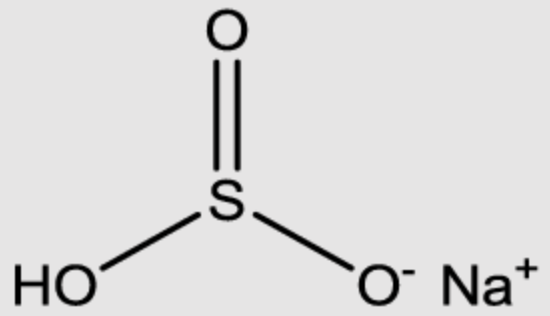
Sodium Bisulfite
Chemical Product
Sodium Bisulfite
Physical and Chemical Properties:
| Properties | |
|---|---|
| Appearance | White crystalline solid |
| Odor | Odorless or slight sulfur odor |
| Molecular Formula | NaHSO3 |
| Molecular Weight | 104.06 g/mol |
| Melting Point | 150 °C |
| Density | 1.48 g/cm³ |
| Solubility | Soluble in water |
| Flash Point | Not applicable |
| Chemical Stability | Stable under normal conditions, but decomposes upon exposure to air or moisture |
Sodium Bisulfite is a chemical compound commonly used as a reducing agent in water treatment, bleaching, and in food preservation.
- Water Treatment: Used as a reducing agent to remove chlorine and to treat water.
- Food Industry: Used as a preservative and antioxidant in food and beverages.
- Chemical Manufacturing: Applied in the production of various chemicals, including sulfur dioxide.
- Photography: Used in the development of photographic film and prints.
Physical and Chemical Properties:
| Properties | Data |
|---|---|
| Appearance | White crystalline solid |
| Odor | Odorless or slight sulfur odor |
| Molecular Formula | NaHSO3 |
| Molecular Weight | 104.06 g/mol |
| Melting Point | 150 °C |
| Density | 1.48 g/cm³ |
| Solubility | Soluble in water |
| Flash Point | Not applicable |
| Chemical Stability | Stable under normal conditions, but decomposes upon exposure to air or moisture |