x
k
l
a
r
c
h
e
m
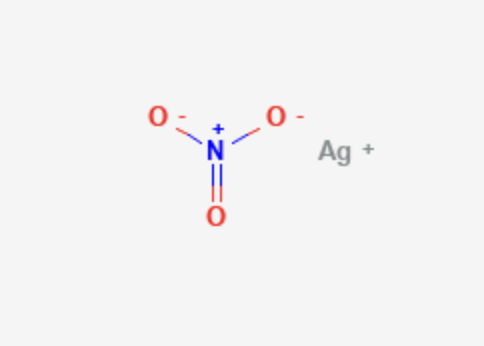
Silver Nitrate
Chemical Product
Silver Nitrate
Physical and Chemical Properties:
| Properties | |
|---|---|
| Appearance | Colorless to white crystalline solid |
| Odor | Odorless |
| Molecular Formula | AgNO3 |
| Molecular Weight | 169.87 g/mol |
| Melting Point | 212 °C |
| Density | 4.35 g/cm³ |
| Solubility | Soluble in water, ethanol, and glycerol |
| Flash Point | Not applicable |
| Chemical Stability | Stable under normal conditions, but decomposes in the presence of light and heat |
Silver Nitrate is an inorganic compound used in chemical analysis, in photography, and as an antimicrobial agent in medical applications.
- Photography: Used in photographic film and paper for the production of images.
- Chemical Manufacturing: Used in chemical reactions, including the synthesis of other silver compounds.
- Medical Applications: Used in wound care and as an antiseptic.
- Analytical Chemistry: Applied in titration and as a reagent in laboratory experiments.
Physical and Chemical Properties:
| Properties | Data |
|---|---|
| Appearance | Colorless to white crystalline solid |
| Odor | Odorless |
| Molecular Formula | AgNO3 |
| Molecular Weight | 169.87 g/mol |
| Melting Point | 212 °C |
| Density | 4.35 g/cm³ |
| Solubility | Soluble in water, ethanol, and glycerol |
| Flash Point | Not applicable |
| Chemical Stability | Stable under normal conditions, but decomposes in the presence of light and heat |