x
k
l
a
r
c
h
e
m
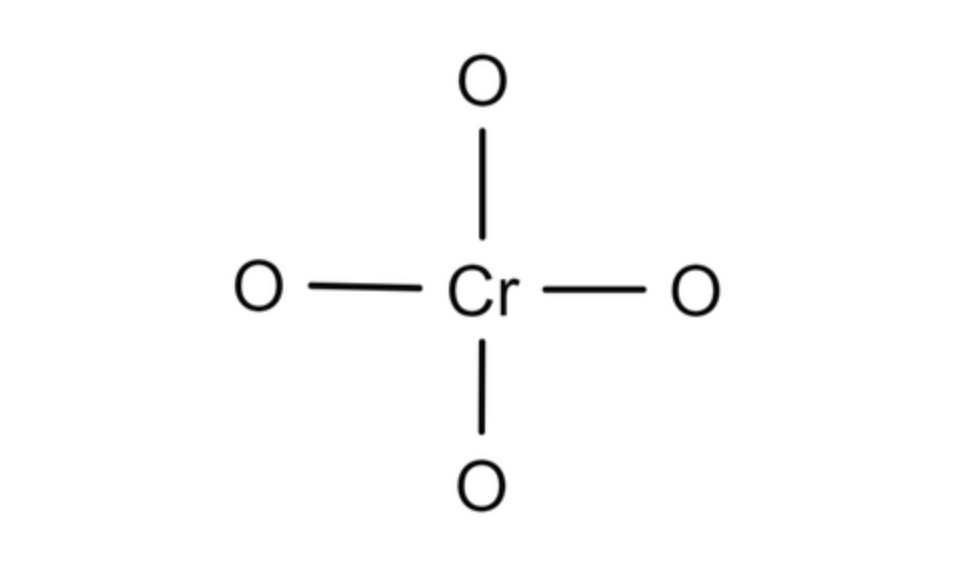
Chromic Acid
Chemical Product
Chromic Acid
Physical and Chemical Properties:
| Properties | |
|---|---|
| Appearance | Red crystalline solid or yellowish liquid |
| Odor | Odorless |
| Molecular Formula | H2CrO4 |
| Molecular Weight | 100.00 g/mol |
| Melting Point | 196 °C (decomposes) |
| Density | 2.52 g/cm³ (at 25 °C) |
| Solubility | Soluble in water |
| pH (1% solution) | <1 (strongly acidic) |
| Chemical Stability | Stable under dry conditions but highly corrosive and reactive in aqueous solutions |
Chromic Acid is a strong oxidizing agent that is highly corrosive. It is commonly used in electroplating, metal cleaning, and in the manufacture of chromium-based chemicals. It is typically available as a concentrated aqueous solution.
- Electroplating: Used for chrome plating on metals.
- Metal Cleaning: Applied in the cleaning and pickling of metals.
- Chemical Manufacturing: Intermediate in the production of chromium chemicals.
- Leather Tanning: Used in leather processing to create durable and flexible leather.
- Laboratory Use: Used in organic synthesis and chemical analysis.
Physical and Chemical Properties:
| Properties | Data |
|---|---|
| Appearance | Red crystalline solid or yellowish liquid |
| Odor | Odorless |
| Molecular Formula | H2CrO4 |
| Molecular Weight | 100.00 g/mol |
| Melting Point | 196 °C (decomposes) |
| Density | 2.52 g/cm³ (at 25 °C) |
| Solubility | Soluble in water |
| pH (1% solution) | <1 (strongly acidic) |
| Chemical Stability | Stable under dry conditions but highly corrosive and reactive in aqueous solutions |