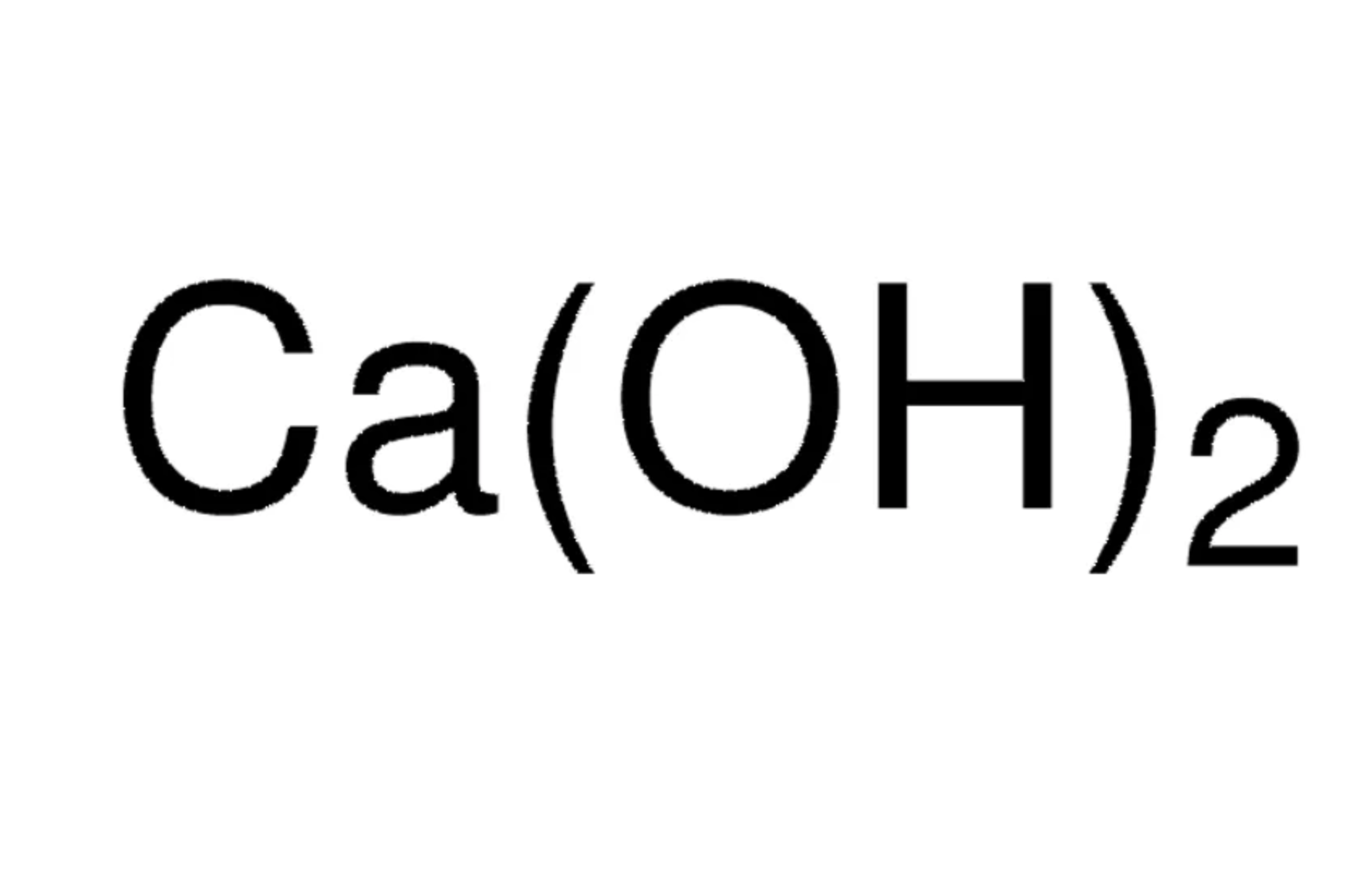x
k
l
a
r
c
h
e
m
Calcium Hydroxide
Chemical Product
Calcium Hydroxide
Physical and Chemical Properties:
| Properties | |
|---|---|
| Appearance | White, odorless powder or crystalline solid |
| Odor | Odorless |
| Molecular Formula | Ca(OH)2 |
| Molecular Weight | 74.09 g/mol |
| Melting Point | Decomposes at 580 °C |
| Density | 2.211 g/cm³ |
| Solubility | Slightly soluble in water, more soluble in acid |
| Flash Point | Not applicable |
| Chemical Stability | Stable under normal conditions, but should be kept dry and away from acids |
Calcium Hydroxide is an inorganic compound commonly used in construction, water treatment, and chemical manufacturing.
- Water Treatment: Used to treat drinking water by neutralizing acids and removing impurities.
- Construction: Applied in the production of mortars, plasters, and cement.
- Agriculture: Used to adjust soil pH and as a fungicide.
- Chemical Manufacturing: Used in the production of calcium compounds and as a pH regulator in various chemical processes.
Physical and Chemical Properties:
| Properties | Data |
|---|---|
| Appearance | White, odorless powder or crystalline solid |
| Odor | Odorless |
| Molecular Formula | Ca(OH)2 |
| Molecular Weight | 74.09 g/mol |
| Melting Point | Decomposes at 580 °C |
| Density | 2.211 g/cm³ |
| Solubility | Slightly soluble in water, more soluble in acid |
| Flash Point | Not applicable |
| Chemical Stability | Stable under normal conditions, but should be kept dry and away from acids |